You are viewing the "Vaccine" Archives
Updated COVID shots are coming. They’re part of a trio of vaccines to block fall viruses
By: Associated Press
Posted on:
WASHINGTON (AP) — Updated COVID-19 vaccines are coming soon, just in time to pair them with flu shots. And this fall, the first vaccines for another scary virus called RSV are rolling… Read More
Why manufacturers are hiking COVID vaccine prices
By: PBS Newshour
Posted on:
WASHINGTON, D.C. (NewsHour) — Since COVID vaccines first became available in the U.S., the federal government has been buying them from manufacturers and distributing them for free. But soon, the… Read More
It’s not too late to get a COVID booster — especially for older adults
By: Selena Simmons-Duffin | NPR
Posted on:
WASHINGTON, D.C. (NPR) — The U.S. has come a long way from two years ago when COVID-19 vaccines first became available and people were cutting the line to get their… Read More
State appeals court revives lawsuit challenging university’s COVID vaccine mandate
By: David Forster
Posted on:
WOUB Digital · State appeals court revives lawsuit challenging university’s COVID vaccine mandate ATHENS, Ohio (WOUB) — A state appeals court has kept alive a lawsuit challenging Miami University’s… Read More
How monoclonal antibodies lost the fight with new COVID variants
By: Pien Huang | NPR
Posted on:
WASHINGTON D.C. (NPR) – Monoclonal antibodies were once the star of COVID-19 outpatient treatments. Since they first became available in 2020 – even before the first vaccines – more than… Read More
Study led by Ohio University professor offers clues to address vaccine resistance in Appalachian Ohio
By: David Forster
Posted on:
WOUB Digital · Study led by Ohio University professor offers clues to vaccine resistance in Appalachian Ohio ATHENS, Ohio (WOUB) — Southeast Ohio as a region has the lowest… Read More

Holzer And O’Bleness Respond To Understaffing And High COVID Numbers With Familiar Pandemic Protocols
By: Kaitlin Thorne
Posted on:
ATHENS, Ohio (WOUB) — COVID-19 numbers continue to rise in Southeast Ohio, causing local health care systems to be stretched thin and re-institute early 2020 pandemic protocols. Holzer Health Systems… Read More

Ohio University Switching COVID-19 Vaccine In Response To Safety Concerns
By: WOUB News Team
Posted on:
ATHENS, Ohio (WOUB) — Ohio University is switching to the Pfizer COVID-19 vaccine for its student clinics after federal officials recommended a temporary halt in use of the Johnson &… Read More
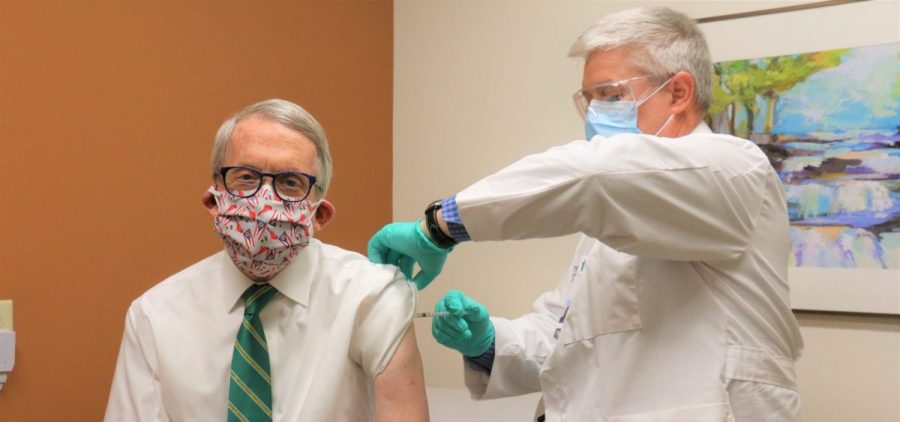
Governor, First Lady Receive 2nd COVID-19 Vaccine Shot
By: Associated Press
Posted on:
COLUMBUS, Ohio (AP) — The office of Ohio Gov. Mike DeWine says the governor and first lady Fran DeWine have received the second dose of the coronavirus vaccine. The Republican… Read More

Ohio Health Officials Tackle COVID-19 Vaccine Myths In Virtual Town Halls
By: Lisa Ryan | WCPN
Posted on:
CLEVELAND, Ohio (WCPN) — The Ohio Department of Health (ODH) wants to combat misinformation about the COVID-19 vaccine by hosting town halls targeting specific, underserved communities and their questions over… Read More

West Virginia’s Vaccination Rate Ranks Among Highest In World
By: Simone Popperl | Noel King | Lisa Weiner | NPR
Posted on:
How did West Virginia become one of the world’s leaders in delivering COVID-19 vaccines? One piece of the story starts with a striking photograph in the local paper.
Ohio To Keep Vaccinations At Age 65+ For ‘Next Few Weeks’
By: Jennifer Merritt | WVXU
Posted on:
COLUMBUS, Ohio (WVXU) — Gov. Mike DeWine on Tuesday said the state will keep its roster of people eligible for the coronavirus vaccine at age 65 and older for the… Read More

DeWine: Ohio Will Have 1,300 Schools Vaccinated By End Of Week
By: Gabe Rosenberg | WOSU
Posted on:
COLUMBUS, Ohio (WOSU) — Calling it an “urgent necessity” to return students to the classroom, Ohio Gov. Mike DeWine says more than 1,300 schools will have their teachers and staff vaccinated… Read More
DeWine: COVID Vaccines Mishandled In 5 NE Ohio Nursing Homes
By: Jennifer Merritt | WVXU
Posted on:
COLUMBUS, Ohio (WVXU) — Some patients at five nursing homes in Northeast Ohio will have to repeat their COVID-19 vaccinations after Walgreens informed the state that some doses weren’t stored… Read More

W.Va. Sees 16% Bump In Vaccine Doses From Federal Government
By: Associated Press
Posted on:
CHARLESTON, W.Va. (AP) — West Virginia officials say President Joe Biden is making good on his pledge to increase vaccine supplies with a 16% bump in first doses received by… Read More

COVID-19 Vaccine: Will It Protect Against New Variants And Do You Need A 2nd Dose?
By: Joanne Silberner | NPR
Posted on:
The spread of new strains raises new questions as two COVID-19 vaccines continue their rollout across the U.S. and another vaccine candidate preps for regulatory review. Here’s what you need to know.
School Teachers, Staff Report Issues Trying To Schedule COVID-19 Vaccine
By: Andy Chow | Statehouse News Bureau
Posted on:
COLUMBUS, Ohio (Statehouse News Bureau) — More than 91,000 school teachers and staff around the state are now eligible for the COVID-19 vaccine. But some educators say they ran into… Read More
Ohio Is Working To Improve COVID Vaccination Scheduling
By: Jo Ingles | Statehouse News Bureau
Posted on:
COLUMBUS, Ohio (Statehouse News Bureau) — If you are over 75 and qualify for a COVID vaccine in Ohio, you can go to the state’s coronavirus website and find places where they… Read More

Ohio’s K-12 School Employees Will Begin Getting COVID-19 Vaccines Thursday
By: Jo Ingles | Statehouse News Bureau
Posted on:
COLUMBUS, Ohio (Statehouse News Bureau) — Ohio’s K-12 schools will begin vaccinating their employees later this week. But the schedule for shot clinics is based on vaccine accessibility. Ohio Gov. Mike… Read More
With COVID Vaccinations Underway, Some Frontline Workers Are Hesitant To Receive It
By: Corinne Boyer | Liam Niemeyer | Ohio Valley ReSource
Posted on:
MURRAY, Ky. (OVR) — More than 969,000 doses of COVID-19 vaccine have been administered to long-term care workers, teachers, and additional frontline health care workers around the Ohio Valley. But … Read More

Virus Vaccine Online Signup Portal Debuts In West Virginia
By: Associated Press
Posted on:
CHARLESTON, W.Va. (AP) — West Virginia is debuting an online portal for residents to register for coronavirus vaccine appointments. Gov. Jim Justice says the new system launches Monday and will… Read More

Trump Administration Had ‘No Plan’ For COVID-19 Vaccine Distribution, Dr. Michael Osterholm Says
By: Robin Young | Samantha Raphelson | Here and Now
Posted on:
The White House says Biden will sign 10 executive orders and directives meant to boost supplies of medical equipment, COVID-19 tests and vaccines.

DeWine Asks Biden For More COVID-19 Vaccine, National Strategies
By: Andy Chow | Statehouse News Bureau
Posted on:
COLUMBUS, Ohio (Statehouse News Bureau) — The transition from a Trump to Biden administration means changes for the nation’s approach to the COVID-19 pandemic. Gov. Mike DeWine (R-Ohio) says there’s one… Read More

Ohioans 80 And Up Eligible For COVID-19 Vaccine Starting This Week
By: Nick Castele | WCPN
Posted on:
Updated: 4:32 p.m., Tuesday, Jan. 19, 2020 COLUMBUS, Ohio (WCPN) —Ohio Gov. Mike DeWine celebrated the next phase of the state’s COVID-19 vaccine rollout on Tuesday, broadcasting the inoculation of… Read More
Biden Administration Will Rename ‘Operation Warp Speed,’ Citing Trump ‘Failures’
By: Merrit Kennedy | NPR
Posted on:
The Biden transition team also announced that Dr. David Kessler will be the White House’s chief science officer of COVID Response.

